- Research
- Open access
- Published:
New risk score for predicting steroid resistance in patients with focal segmental glomerulosclerosis or minimal change disease
Clinical Proteomics volume 17, Article number: 18 (2020)
Abstract
Background
Glucocorticosteroid is used for patients with primary nephrotic syndrome. This study aims to identify and validate that biomarkers can be used to predict steroid resistance.
Methods
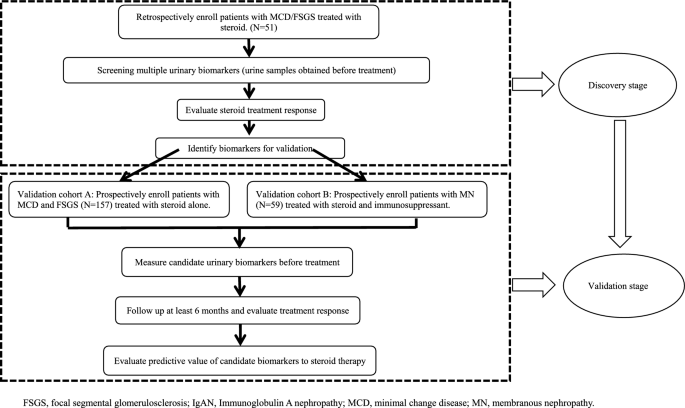
Our study contained two stages, discovery and validation stage. In discovery stage, we enrolled 51 minimal change disease (MCD) or focal segmental glomerulosclerosis (FSGS) patients treated with full dose steroid. Five urinary biomarkers including β2-microglobulin (β2-MG) and α1-microglobulin (α1-MG) were tested and candidates’ biomarkers were selected based on their associations with steroid response. In validation stage, candidates’ biomarkers were validated in two prospectively enrolled cohorts. Validation cohort A included 157 FSGS/MCD patients. Validation cohort B included 59 membranous nephropathy (MN) patients. Patients were classified into response group (RG) or non-response group (NRG) based on their responses to steroid treatment.
Results
In discovery stage, higher urinary β2-MG was independently associated with response to corticosteroid treatment in MCD/FSGS patients [OR = 1.89, 95% CI 1.02–3.53] after adjusted by age and gender. In validation cohort A, patients in NRG had a significant higher urinary β2-MG [Ln (β2-MG/uCr): 4.6 ± 1.7 vs 3.2 ± 1.5] compared to patients in RG. We then developed a 3-variable risk score in predicting steroid resistance in FSGS/MCD patients based on the best predictive model including Ln(β2-MG/uCr) [OR = 1.76, 95% CI 1.30–2.37], age [OR = 1.005, 95% CI 0.98–1.03] and pathology [MCD vs FSGS, OR = 0.20, 95% CI 0.09–0.46]. The area under the ROC curves of the risk score in predicting steroid response was 0.80 (95% CI 0.65–0.85). However, no such association was found in MN patients.
Conclusions
Our study identified a 3-variable risk score in predicting steroid resistance in patients with FSGS or MCD.
Background
Primary glomerulonephritis including primary nephrotic syndrome (PNS) is the most common cause of end stage renal disease (PG) in China. Based on pathological changes, common types of PNS include focal segmental glomerulosclerosis (FSGS), minimal change disease (MCD) and membranous nephropathy (MN). The mechanism of PNS is still obscure although some major progresses have been made, such as the findings of PLA2R and THSD7A in MN, Gd-IgA1 in IgAN and podocyte-related genes such as INF2 and APOL1 in FSGS [1,2,3,4]. However, no specific agents are available for the treatment of PNS as of today. Therefore, corticosteroids and immunosuppressants are still widely used when massive proteinuria occurs despite the following constraints. First, a significant proportion of these patients show poor responses to the medication. Furthermore, and severe side effects might occur such as infection, metabolic disturbance or osteoporosis [5, 6]. Various risk factors were found to be associated with steroid resistance, including age, abnormal expression of glucocorticoid receptor, mutations of podocyte-related genes, pathological types, abnormal lipid metabolism or immune factors [7]. Given the drawbacks mentioned above, predicting patients’ response before steroid treatment can be very useful. Unfortunately, there is no clinically applicable method to achieve this goal as of now.
Recently, several studies have focused on predictive value of urinary biomarkers to steroid resistance; however, the results of these biomarkers were uncertain and need to be further validated. Five selected biomarkers were illustrated in our study. The first among them were β2-microglobulin (β2-MG), a low-molecular-weight protein (11 kDa) [8, 9] and a single-chain polypeptide consisting of 99 amino acids, a component of human leukocyte antigen (HLA) β chain (light chain) produced by lymphocytes, platelets or polymorphonuclear leukocytes. Hofstra’s study [10] included 57 patients with membranous nephropathy. They found patients with lower urinary β2-MG had a higher remission rate. Therefore, they concluded that urinary β2-MG levels were useful in predicting prognosis. The second biomarker, α1-microglobulin (α1-MG), is another low-molecular-weight protein (26–32 kDa) [11] which is mainly synthesized by liver and lymphocytes. α1-MG is comprised of 167 amino acids and crossreacts with antigen determinants such as HLA. Studies [12] showed that the increase of α1-MG reflected early renal tubulointerstitial injuries. The third urinary biomarker- orosomucoid (ORM), with a molecular weight of approximately 40,000 Da, is mainly produced in the liver in the form of a single-chain polypeptide with five multi-branched N-sugar chains [13]. Previous studies demonstrated that plasma orosomucoid increased in response to inflammation and other stressful stimuli. A few studies [14, 15] showed that urinary excretion of orosomucoid (UOER) was very low in healthy people. Other studies [16, 17] discovered that increased UOER was an independent, powerful predictor of cardiovascular mortality in patients with type 2 diabetes and diabetic nephropathy. The fourth urinary biomarker, yet a frequently detected one is microalbumin (MAU). MAU presenting in patients with type 2 diabetes indicated poorer renal outcomes and increased risk for ESRD [18]. In addition, the urinary microalbumin creatinine ratio is an early and reliable biomarker for renal injury [19, 20]. The last biomarker is retinol binding protein (RBP), a protein with molecular weight of 21,200 Da [21]. RBP consists of a polypeptide chain and a small portion of carbohydrates and it is mainly produced by liver cells and widely distributed in serum, cerebrospinal fluid, urine and other body fluids. RBP was proved to be a biomarker for interstitial fibrosis [22].
This study aims to identify and validate urinary biomarkers that can predict the response to steroid treatment in PNS patients and to establish a risk score to predict steroid response by combine the effect of potential biomarkers and clinical variables.
Materials and methods
Study population and design
Our study contained two stages (Fig. 1). In the discovery stage, biopsy-proven PNS patients including FSGS or MCD were retrospectively enrolled. The inclusion criteria included PNS patients with biopsy-proven FSGS or MCD; urine protein excretion more than 3.5 g/day; and serum albumin less than 30 g/L. The exclusion criteria were secondary glomerulonephritis such as systemic lupus erythematosus, renal amyloidosis, obesity-related nephropathy, diabetic nephropathy, systemic vasculitis, and human immunodeficiency virus (HIV) related kidney disease; and pregnancy. All patients were given full dosage of prednisone or prednisolone alone [1 mg/(kg/day), maximum to 80 mg/day].
In the validation stage, biopsy-proven PNS patients were enrolled and divided into three cohorts depending on their pathologic diagnosis including FSGS or MCD (validation cohort A) or MN (validation cohort B). The inclusion criteria included patients with biopsy-proven PNS; urine protein excretion more than 3.5 g/day; and serum albumin less than 30 g/L. The exclusion criteria were secondary glomerulonephritis, such as systemic lupus erythematosus, renal amyloidosis, obesity-related nephropathy, diabetic nephropathy, systemic vasculitis, and human immunodeficiency virus (HIV) related kidney disease; and pregnancy. Patients were given full dosage of prednisone/prednisolone alone [1 mg/(kg/day), maximum to 80 mg/day] in validation cohort A or prednisone/prednisolone [0.5–0.8 mg/(kg/day)] combined with immunosuppressants in validation cohort B. Immunosuppressants included cyclophosphamide (0.5 g/m2 every month), Cyclosporine (3–4 mg/kg/day), Tacrolimus (0.05 mg/kg/day) or mycophenolate mofetil (1–2 g/day).
The primary outcome of this study was proteinuria remission, which was defined as urine protein decreased by more than 50% compared to baseline and was less than 3.5 g/day after 8 weeks of prednisone/prednisolone treatment. Patients were classified as the response group (RG) or non-response group (NRG) based on whether the primary outcome was achieved.
Baseline characteristics
Gender, age, body weight and height, systolic blood pressure (SBP), diastolic blood pressure (DBP), creatinine (Scr), urine protein, albumin, body mass index (BMI), uric acid (UA); total cholesterol (TG), and total cholesterol [23] were recorded at the time of renal biopsy. Urine was collected in the morning after at least 9 h’ water prohibition. Urinary α1-MG was measured by rate nephelometry (BNII Protein Analyzer, Siemens, Germany). Urinary β2-MG was measured by chemiluminescent technique (IMMULITE 2000, Siemens, Germany). Urinary ORM and MAU were measured by Immunoturbidimetry (Dako, Denmark). Urinary RBP was measured by Immunoturbidimetry (Byron, Shanghai). Estimated GFR [24] was calculated using the CKD-EPI formula (2009) [25]. Patients were followed up for at least 6 months after recruitment.
Histological examination
All biopsy specimens were processed by the standard methods for histological examination. Specimens were stained with hematoxylin–eosin, Masson’s trichrome and methenamine silver. Pathologic changes were scored by semiquantitative pathological evaluation with details as below. Global sclerosis was defined as sclerosis involving the entire glomerular tuft. Global sclerosis was scored by the presence of glomeruli with these lesions: 0, absent; 1, present. Segmental sclerosis was defined as tufts involved with sclerosis other than global sclerosis. Segmental sclerosis was scored as followed: 0, absent; 1, present. The severity of interstitial fibrosis, interstitial inflammatory cell infiltration and tubular atrophy were defined as followed: 0, absent; 1, present. The vascular lesion was defined by arterial hyaline change and vascular wall thickening: for either, definitions were 0, absent, or 1, present.
Statistical analysis
All statistics were performed using SPSS software (version 22). Values are presented as the mean ± standard deviation (SD), unless otherwise stated. As α1-MG and β2-MG levels did not follow a normal distribution, they were normalized by natural logarithm. Independent T test was employed to continuous variables conformed to the normal distribution to evaluate the significance of the difference of mean values between two groups. The variables included age, MAP, BMI, and normal transformed variables including albumin, eGFR, TG, TC, urine protein and β2-MG by natural logarithm. Categorical variables such as pathological evaluation were expressed as frequencies or percentages (%). Pearson Chi square test was applied. We also used logistic regression analysis for evaluating associations between urinary biomarkers and treatment response. In addition, areas under receiver operating characteristic [26] curves were also assessed, and the best diagnostic threshold values were selected. Differences were considered statistically significant with a two-side P value < 0.05.
Results
Demographic data and clinical features of patients in the discovery stage
A total of 51 patients were enrolled in the discovery cohort. There were 11 patients with FSGS (21.6%) and 40 with MCD (78.4%). Of all patients, 10 (19.6%) were in the NRG and 41 (81.4%) were in the RG. There were no differences among demographic and clinical variables such as gender, age, urine protein and eGFR between patients from RG and NRG (Table 1).
Screening of urinary biomarkers in the discovery stage
In all patients in the discovery stage, baseline urinary Ln(β2-MG) (6.6 ± 1.8 vs 5.7 ± 1.4) was significantly increased in NRG versus RG (P < 0.01). However, urinary MAU (7.9 ± 1.3 vs 8.5 ± 1.2, P > 0.05), ORM (5.0 ± 1.2 vs 5.6 ± 1.0, P > 0.05), RBP (− 1.1 ± 1.7 vs − 0.7 ± 1.7, P > 0.05) and urinary Ln(α1-MG) (1.6 ± 0.9 vs 1.3 ± 1.0, P > 0.05) showed no difference between the two groups.
Demographic data and clinico-histological features of patients in the validation stage
In total, 216 biopsy-proven patients were enrolled and divided into two cohorts based on their pathologic diagnosis including 157 FSGS and MCD in validation cohort A and 59 MN in validation cohort B. Patients were given full dosage of prednisone/prednisolone alone [1 mg/(kg/day)] in validation cohort A or prednisone/prednisolone [0.5–0.8 mg/(kg/day)] combined with immunosuppressants in validation cohort B. Among patients from validation cohort B, 24 patients used cyclophosphamide (CTX, 0.5 g/m2 every month), 23 patients used Cyclosporine (3–4 mg/kg/day) and eight patients used Tacrolimus (0.05 mg/kg/day). Of all patients in validation cohort A, 54 patients were in NRG (34.4%) and 103 were in RG (65.6%) (Table 2). Baseline eGFR (81.9 ± 37.5 vs 99.1 ± 27.5) (P < 0.01) was significantly decreased in NRG versus RG (OR = 0.98, 95% CI 0.97–0.99). Compared to the RG, patients in the NRG had higher ratios of severe global sclerosis (OR = 2.25, 95% CI 1.11–4.56, P < 0.05), segmental sclerosis (OR = 5.49, 95% CI 2.47–12.17, P < 0.01), interstitial fibrosis (OR = 3.83, 95% CI 1.63–8.97, P < 0.01), inflammatory cell infiltration (OR = 2.20, 95% CI 1.06–4.56, P < 0.05) and tubular atrophy (OR = 2.60, 95% CI 1.22–5.55, P < 0.05) (Table 2).
Validating of urinary β2-MG with response to steroid treatment in the validation stage
Then we validate the urinary β2-MG in the validation cohort. We confirmed baseline urinary Ln(β2-MG/uCr) (4.6 ± 1.7 vs 3.2 ± 1.5, P < 0.01) was significantly increased in NRG versus RG in validation cohort A. Univariate logistic regression analysis showed higher urinary Ln(β2-MG/uCr) (OR = 1.76, 95% CI 1.30–2.37, P < 0.01) was associated with higher risk for steroid resistance in FSGS and MCD patients (Table 2). Even after fully adjusting for age, gender, clinical parameters and pathological parameters (segmental sclerosis, interstitial fibrosis, inflammatory cell infiltration and tubular atrophy), higher urinary Ln(β2-MG/uCr) (OR = 1.51, 95% CI 1.09–2.08, P < 0.01) was found to be independently associated with steroid resistance in FSGS and MCD patients. However, the associations between Ln(β2-MG/uCr) and steroid resistance were not validated in patients with MN (validation cohort B) (OR = 1.51, 95% CI 0.36–6.38) (Tables 3 and 4).
In the multivariate stepwise logistic regression model, the best model in predicting steroid resistance included three baseline variables (Fig. 2). These included: Ln(β2-MG/uCr) [OR = 1.76, 95% CI 1.30–2.37], age [OR = 1.005, 95% CI 0.98–1.03], pathology (defined as FSGS = 1, MCD = 3) [OR = 0.20, 95% CI 0.09–0.46]. Based on this model, we developed a 3-variable equation to calculate risk score for predicting steroid resistance in FSGS/MCD patients. The discrimination of the risk score was estimated by AUC (the area under the ROC curves) in the validation cohort A [0.80 (0.65–0.85)], indicating good discrimination (Fig. 2).
Furthermore, the associations between urinary Ln(β2-MG/uCr) and steroid resistance were robust in subgroups analysis, including eGFR and some pathological parameters subgroups, except in elderly and overweight patients (Fig. 3). We divided patients into two tertile groups according to their urinary β2-MG value. Clinico-histological features and response to steroid treatment were compared among two groups. Compared to patients in the low tertile group, patients in the high tertile group had lower eGFR and higher levels of TG and tubular-interstitial lesion. No response rate was significantly higher in patients from the high tertile group (OR = 11.38, 95% CI 3.94–32.84, P < 0.01) compared with patients from the low tertile group (Table 5).
Discussion
Corticosteroids remain to be used as the basic treatment of PNS patients in spite of the emerging treatments and drugs in recent years. Past clinical practice of steroid application has indicated that steroid treatment has been proved to be effective for some PNS patients especially those with MCD/FSGS, whereas other patients are steroid resistance. Of those patients who respond to steroid at the initial stage of treatment, some cease to respond effectively as the disease progresses. The fact that PNS patients respond differently to steroid therapy has raised the concern that serious complications resulted from large doses of steroid usage could have happened if patients were to show steroid resistance during treatment. Unfortunately, clinicians are still unable to predict steroid resistance before treatment. Therefore, identification of risk factors for steroid resistance will be essential to clinical practice to reduce the side effects of unnecessary corticosteroid therapy. In this study, we first tested five urinary biomarkers from both FSGS and MCD patients in the discovery cohort and then recorded and identified their associations with steroid resistance. One candidate urine biomarker, urinary β2-MG, was selected and validated in the three validation cohorts. In summary, we identified and validated that urinary β2-MG could be used as an early biomarker for steroid resistance in patients with FSGS or MCD.
A previous study [5] showed that steroid resistance was related to the renal pathological types of patients. For instance, most MCD patients are steroid sensitive, while approximately 30–50% of FSGS patients are steroid resistant [6]. In our study, we found that 42.3% FSGS patients presented with steroid resistance, higher than MCD patients (21.4%). We therefore concluded that the data was aligned with that of the previous study. Increasing number of studies have shown that certain biomarkers could be utilized as important reference for clinical work because of their potential values in predicting the effect of steroid therapy and prognosis of PNS. However, the conclusions of these studies still raise concerns given the constraints that no systematic investigation of different urinary biomarkers was conducted and most of the study results lacked external validation. Our study investigated and validated the predictive value of a series of urinary biomarkers in PNS patients. In the discovery stage, we found that baseline urinary β2-MG significantly increased in FSGS and MCD patients in the steroid resistant group versus those in the steroid sensitive group. Then, the finding was validated in a prospectively collected FSGS and MCD cohort, and an independent association between higher urinary β2-MG and steroid resistance was successfully validated. No such relationship was found in MN patients.
β2-microglobulin, a light-chain molecule of the major histocompatibility complex (MHC) class I antigens, is one of these proteins we screened. β2-microglobulin is a low-molecular-weight protein that is found on the surface membrane of almost all nucleated cells. β2-microglobulin is freely filtered by the glomerular filtration barrier and reabsorbed by the renal tubular cells. β2-MG levels increase in patients with diseases such as renal tubulointerstitial injuries, autoimmune disease and multiple myeloma [27]. Previous studies showed that the level of urinary β2-MG and N-acetyl-beta-d-glucosaminidase (NAG) associated with interstitial-tubular lesions were elevated in steroid resistant patients. In the study of Fede et al. [28], urinary NAG and β2-MG levels were assessed in 19 patients with nephrotic syndrome (NS) and in 30 healthy controls. Results showed that steroid resistant patients had significant higher levels of baseline urinary NAG and β2-MG levels than steroid sensitive patients and healthy controls. In the study of Vallés et al. [29], 34 patients with NS were enrolled. Urinary β2-MG level was significantly increased in patients with steroid resistant NS (SRNS) compared to patients with steroid dependent NS (SDNS) or steroid sensitive NS (SSNS). There were no differences between the SRNS group and SSNS in relapse. Urinary NAG level was significantly higher in patients in the SRNS group than those in the SDNS, SSNS, and control groups. However, it was still unclear whether the association with β2-MG, NAG and steroid resistance was independent of interstitial-tubular lesions. Sesso et al. [30] enrolled 37 patients with the nephrotic syndrome. They suggested β2-MG levels could help identify patients more sensitive to steroids. Although several studies can be found about urinary β2-MG and steroid resistance in renal diseases, none of these studies focused on FSGS and MCD which had unique pathogenesis compared to other kidney diseases. In addition, limited sample size restrained these studies from excluding the effects of other confounding factors such as tubulointerstitial lesions. In the present study, we found urinary β2-MG level of FSGS and MCD patients was associated with interstitial fibrosis, inflammatory cell infiltration and tubular atrophy, and increased with the severity of renal tubular injury. Nonetheless, the association between urinary β2-MG and steroid response was robust after adjusting for demographic, clinical and histological variables including segmental sclerosis, interstitial fibrosis, inflammatory cell infiltration and tubular atrophy. Furthermore, subgroup analysis showed urinary β2-MG level was significantly associated with steroid resistance. The conclusion still held true, even for patients without inflammatory cell infiltration. Therefore, our data indicates that the association between urinary β2-MG and corticosteroids response not only reflects tubulointerstitial injury but also suggests an additional mechanism linked to steroid resistance.
β2-MG levels are mainly determined by turnover and activity of lymphocytes in lymphoproliferative and autoimmune diseases [26, 31, 32] such as in Sjögren’s syndrome and SLE. Prior to recognizing the important role that antiphospholipase A2 receptor (anti-PLA2R) autoantibodies played in primary MN, consistent data from studies in animals had already shown that antibody produced by autoreactive B cell clones initiated the events that eventually led to injury to the glomerular filtration barrier and consequent proteinuria [2, 33,34,35]. The data above suggested that activation of B cells might result in the increase of serum β2-MG level. Other studies had shown that increase of serum β2-MG level would be the consequence of an enhanced surface expression and shedding of MHC class I molecules, mainly on activated macrophages induced by various cytokines (IFN-γ, TNF-α, etc.) [31]. These studies implied that activation of B cells and macrophages might contribute to the increase of serum β2-MG level. Further research is needed to clarify the mechanism.
We acknowledge that this study has limitations. First, in the discovery stage, although urine was collected in the morning after water prohibition, urine proteins were not adjusted by urinary creatinine. Second, multicenter clinical trials with large sample sizes are necessary to validate our findings given the limitation of our single center study. Lastly, combined effects of multiple biomarkers may better enhance the creditability and accuracy in predicting corticosteroid response than the effect of one single biomarker in our study.
Conclusions
In short, our study screened five urinary biomarkers PNS patients. We identified and validated that urinary β2-MG could independently predict the response to corticosteroid treatment. We developed a 3-variable risk score in predicting steroid resistance in patients with FSGS or MCD. Our results are helpful for clinicians to avoid the abuse of steroid usage in these patients to decrease unnecessary side effects.
Availability of data and materials
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- FSGS:
-
Focal segmental glomerulosclerosis
- IgAN:
-
Immunoglobulin A nephropathy
- MCD:
-
Minimal change disease
- RG:
-
Response group
- NRG:
-
Non-response group
- MAP:
-
Mean arterial pressure
- BMI:
-
Body mass index
- Ln(Pro):
-
Natural logarithm of urine protein
- eGFR:
-
Estimated glomerular filtration rate
- TG:
-
Triglyceride
- TC:
-
Total cholesterol
- MAU:
-
Microalbumin
- ORM:
-
Orosomucoid
- RBP:
-
Retinol binding protein
- β2-MG:
-
β2-Microglobulin
- α1-MG:
-
α1-Microglobulin
References
Tomas NM, Beck LH Jr, Meyer-Schwesinger C, Seitz-Polski B, Ma H, Zahner G, et al. Thrombospondin type-1 domain-containing 7A in idiopathic membranous nephropathy. N Engl J Med. 2014;371(24):2277–87.
Beck LH, Bonegio RGB, Lambeau G, Beck DM, Powell DW, Cummins TD, et al. M-Type phospholipase A2 receptor as target antigen in idiopathic membranous nephropathy. N Engl J Med. 2009;361(1):11–21.
Gharavi AG, Moldoveanu Z, Wyatt RJ, Barker CV, Woodford SY, Lifton RP, et al. Aberrant IgA1 glycosylation is inherited in familial and sporadic IgA nephropathy. J Am Soc Nephrol. 2008;19(5):1008–14.
Suzuki H. Biomarkers for IgA nephropathy on the basis of multi-hit pathogenesis. Clin Exp Nephrol. 2019;23(1):26–31.
Haas M, Meehan SM, Karrison TG, Spargo BH. Changing etiologies of unexplained adult nephrotic syndrome: a comparison of renal biopsy findings from 1976–1979 and 1995-1997. Am J Kidney Dis. 1997;30(5):621–31.
Sasaki TM, Pirsch JD, D’Alessandro AM, Knechtle SJ, Kalayoglu M, Hafez GR, et al. Increased beta 2-microglobulin (B2M) is useful in the detection of post-transplant lymphoproliferative disease (PTLD). Clin Transpl. 1997;11(1):29–33.
Li Y, He Q, Wang Y, Dang X, Wu X, Li X, et al. A systematic analysis of major susceptible genes in childhood-onset steroid-resistant nephrotic syndrome. Ann Clin Lab Sci. 2019;49(3):330–7.
Choe JY, Park SH, Kim SK. Urine β2-microglobulin is associated with clinical disease activity and renal involvement in female patients with systemic lupus erythematosus. Lupus. 2014;23(14):1486–93.
Cristofano C, Vernaglione L, Perniola MA, Lo CB, Muscogiuri P, Chimienti S. Cystatin C, beta2-microglobulin and C-reactive protein in hemodiafiltration and on-line endogenous liquid reinfusion and in low flux polysulphone bicarbonate conventional hemodialysis]. Giornale Italiano di Nefrologia. 2004;21:S197–200.
Hofstra JM, Deegens JKJ, Willems HL, Wetzels JFM. Beta-2-microglobulin is superior to N-acetyl-beta-glucosaminidase in predicting prognosis in idiopathic membranous nephropathy. Nephrol Dial Transpl August. 2008;23(8):2546–51.
Donaldson MD, Chambers RE, Woolridge MW, Whicher JT. Alpha 1-microglobulin, beta 2-microglobulin and retinol binding protein in childhood febrile illness and renal disease. Pediatr Nephrol. 1990;4(4):314–8.
Penders J, Delanghe JR. Alpha 1-microglobulin: clinical laboratory aspects and applications. Clin Chim Acta. 2004;346(2):107–18.
Trubetskaia OE, Belogrudov GI, Danilov AV, Titeeva GR, Afanas’eva GV, Muranova TA, et al. Analysis of the trypsin hydrolysate of a 40-kDa protein, found in the nucleoprotein of serum. Identification of it as alpha(1)-acidic glycoprotein (orosomucoid). Bioorg Khim. 1991;17(1):47–52.
Schmid K. Human plasma alpha 1-acid glycoprotein–biochemical properties, the amino acid sequence and the structure of the carbohydrate moiety, variants and polymorphism. Prog Clin Biol Res. 1989;300:7–22.
Logdberg L, Wester L. Immunocalins: a lipocalin subfamily that modulates immune and inflammatory responses. Biochim Biophys Acta. 2000;1482(1–2):284–97.
Christiansen MS, Hommel E, Magid E, Feldt-Rasmussen B. Orosomucoid in urine is a powerful predictor of cardiovascular mortality in normoalbuminuric patients with type 2 diabetes at five years of follow-up. Diabetologia. 2005;48(2):386–93.
Jiang H, Guan G, Zhang R, Liu G, Liu H, Hou X, et al. Increased urinary excretion of orosomucoid is a risk predictor of diabetic nephropathy. Nephrology. 2009;14(3):332–7.
Abbate M, Zoja C, Remuzzi G. How does proteinuria cause progressive renal damage? J Am Soc Nephrol. 2006;17(11):2974–84.
Keane WF, Eknoyan G. Proteinuria, albuminuria, risk, assessment, detection, elimination (PARADE): a position paper of the National Kidney Foundation. Am J Kidney Dis. 1999;33(5):1004–10.
Karar T, Alniwaider RA, Fattah MA, Al Tamimi W, Alanazi A, Qureshi S. Assessment of microalbuminuria and albumin creatinine ratio in patients with type 2 diabetes mellitus. J Nat Sci Biol Med. 2015;6:S89–92.
Liu KH, Huang JC, Godkin JD. Characterization of protein production by caprine placental membranes: identification and immunolocalization of retinol-binding protein. J Endocrinol. 1995;146(3):527–34.
Pallet N, Chauvet S, Chasse JF, Vincent M, Avillach P, Levi C, et al. Urinary retinol binding protein is a marker of the extent of interstitial kidney fibrosis. PLoS ONE. 2014;9(1):e84708.
Li M, O’Sullivan KM, Jones LK, Semple T, Kumanogoh A, Kikutani H, et al. CD100 enhances dendritic cell and CD4 + cell activation leading to pathogenetic humoral responses and immune complex glomerulonephritis. J Immunol. 2006;177(5):3406–12.
Leng S, Weissfeld JL, Picchi MA, Styn MA, Claus ED, Clark VP, et al. A prospective and retrospective analysis of smoking behavior changes in ever smokers with high risk for lung cancer from New Mexico and Pennsylvania. Int J Mol Epidemiol Genet. 2016;7(2):95–104.
Levey AS, Stevens LA, Schmid CH, Zhang YL, Castro AF 3rd, Feldman HI, et al. A new equation to estimate glomerular filtration rate. Ann Intern Med. 2009;150(9):604–12.
Gottenberg JE, Seror R, Miceli-Richard C, Benessiano J, Devauchelle-Pensec V, Dieude P, et al. Serum levels of beta2-microglobulin and free light chains of immunoglobulins are associated with systemic disease activity in primary Sjogren’s syndrome Data at enrollment in the prospective ASSESS cohort. PLoS ONE. 2013;8(5):e59868.
Bokenkamp A, Grabensee A, Stoffel-Wagner B, Hasan C, Henne T, Offner G, et al. The beta2-microglobulin/cystatin C ratio–a potential marker of post-transplant lymphoproliferative disease. Clin Nephrol. 2002;58(6):417–22.
Fede C, Conti G, Chimenz R, Ricca M. N-acetyl-beta-D-glucosaminidase and beta2-microglobulin: prognostic markers in idiopathic nephrotic syndrome. J Nephrol. 1999;12(1):51–5.
Valles P, Peralta M, Carrizo L, Martin L, Principi I, Gonzalez A, et al. Follow-up of steroid-resistant nephrotic syndrome: tubular proteinuria and enzymuria. Pediatr Nephrol. 2000;15(3–4):252–8.
Sesso R, Santos AP, Nishida SK, Klag MJ, Carvalhaes JT, Ajzen H, et al. Prediction of steroid responsiveness in the idiopathic nephrotic syndrome using urinary retinol-binding protein and beta-2-microglobulin. Ann Intern Med. 1992;116(11):905–9.
Wakabayashi K, Inokuma S, Matsubara E, Onishi K, Asashima H, Nakachi S, et al. Serum beta2-microglobulin level is a useful indicator of disease activity and hemophagocytic syndrome complication in systemic lupus erythematosus and adult-onset Still’s disease. Clin Rheumatol. 2013;32(7):999–1005.
Bethea M, Forman DT. Beta 2-microglobulin: its significance and clinical usefulness. Ann Clin Lab Sci. 1990;20(3):163–8.
Pescovitz MD. Rituximab, an anti-cd20 monoclonal antibody: history and mechanism of action. Am J Transplant. 2006;6(5 Pt 1):859–66.
Cravedi P, Remuzzi G, Ruggenenti P. Rituximab in primary membranous nephropathy: first-line therapy, why not? Nephron Clin Pract. 2014;128(3–4):261–9.
Debiec H, Ronco P. Immunopathogenesis of membranous nephropathy: an update. Semin Immunopathol. 2014;36(4):381–97.
Acknowledgements
This work was supported by grants from the National Key Research and Development Program of China (2016YFC0904100), National Natural Science Foundation of China (Nos. 81870460, 81570598, 81370015), Science and Technology Innovation Action Plan of Shanghai Science and Technology Committee (No. 17441902200), Shanghai Municipal Education Commission, Gaofeng Clinical Medicine Grant (No. 20152207), Shanghai Jiao Tong University School of Medicine, Multi-Center Clinical Research Project (No. DLY201510), Shanghai Health and Family Planning Committee Hundred Talents Program (No. 2018BR37).
Funding
Not applicable.
Author information
Authors and Affiliations
Contributions
Research idea and study design: JX, NC, QW, QZ, XP, HR, WW; data acquisition: QW, QZ, XY; statistical analysis: QW, QZ, JX; data interpretation: XJ, JY, LY, TJ. Each author contributed important intellectual content during manuscript drafting or revision and accepts accountability for the overall work by ensuring that questions pertaining to the accuracy or integrity of any portion of the work are appropriately investigated and resolved. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
All procedures performed in studies involving human participants were in accordance with the ethical standards of the Institutional Review Board of Ruijin Hospital, Shanghai Jiao Tong University School of Medicine and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. The ethics approval number is RJ2012LinLunNo39.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Weng, Q., Zhou, Q., Tong, J. et al. New risk score for predicting steroid resistance in patients with focal segmental glomerulosclerosis or minimal change disease. Clin Proteom 17, 18 (2020). https://doi.org/10.1186/s12014-020-09282-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12014-020-09282-x